MedTech News
.................... by Andrew Celentano

AI model detects normally ‘invisible’ tissue changes of pancreatic cancer at stage 0
An AI model (REDMOD) can pick up the very early subtle tissue changes of pancreatic ductal adenocarcinoma, the most common form of pancreatic cancer, which conventional imaging and the human eye find difficult to detect, finds research published online in the journal Gut.
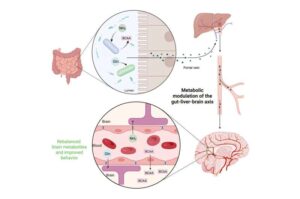
From gut to brain: Scientists engineer bacteria to treat severe liver-related brain dysfunction
A research team engineered strains of a naturally occurring beneficial gut bacterium to function as programmable therapeutics capable of restoring metabolic balance across the gut, liver and brain.
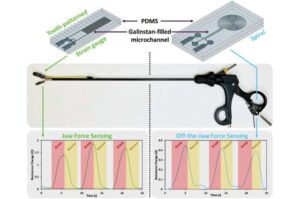
Smart soft sensors restore surgeons’ sense of touch in minimally invasive procedures
Researchers at NYU Abu Dhabi have developed soft, flexible sensors that help restore a surgeon’s sense of touch during minimally invasive (keyhole) surgery.
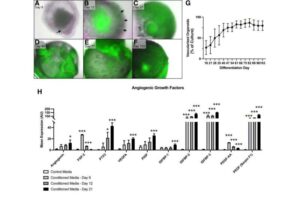
Lab-grown human skin advances our understanding of the critical role of skin blood vessels
New research has shown that single blood vessel cells that appear in the earliest stages of lab-grown skin organoids have the ability to form complex microvascular networks that grow and mature over time.
3Shape Receives FDA 510(k) Clearance for 3Shape Dx Software
COPENHAGEN, Denmark, April 28, 2026 /PRNewswire/ — 3Shape today announced that the U.S. Food and Drug Administration (FDA) has granted FDA clearance for its Dx Software following a review completed on April 10. 2026.
U.S. FDA Grants Breakthrough Device Designation to Laguna Diagnostics’ mRNA Gene Biomarker Test to Aid in Differentiation of Schizophrenia and Bipolar I Disorder
IRVINE, Calif., April 28, 2026 /PRNewswire/ — Laguna Diagnostics, LLC today announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation to the company’s mRNA Gene Biomarker Test, a novel blood-based test designed to aid in the differentiation of schizophrenia and bipolar I disorder in symptomatic patients.
Lasix® ONYU[1] Achieves CE Marking under EU Medical Device Regulation
BURLINGTON, Mass., April 28, 2026 /PRNewswire/ — SQ Innovation, developer of Lasix® ONYU, today announced two important regulatory milestones in its international expansion strategy: the receipt of a CE marking certificate for the Lasix ONYU device, and the acceptance of Lasix ONYU by the European Medicines Agency (EMA) for review under the centralized procedure on the basis of technical innovation. Together, these milestones represent a significant step forward in bringing this novel at-home subcutaneous furosemide treatment to patients in Europe and other international markets.

Motif Neurotech wins FDA IDE to evaluate brain implant for treating depression
Motif Neurotech announced today that it received FDA investigational device exemption (IDE) to evaluate its brain stimulation implant.
