MedTech News
.................... by Andrew Celentano
Recor Medical also secures renal denervation reimbursement in Japan
Recor Medical is gaining reimbursement in Japan’s healthcare system for its Paradise renal denervation for treating hypertension, enabling it to better compete with Medtronic’s rival Symplicity Spyral system.

Olympus picks up pediatric indications from FDA for duo of single-use bronchoscopes
Olympus announced today that it received FDA clearance for pediatric use for two of the single-use bronchoscopes it distributes in the U.S.

Haemonetics Receives FDA Clearance for NexSys® PCS Plasma Collection System with Persona® PLUS Technology
BOSTON, Feb. 23, 2026 /PRNewswire/ — Haemonetics Corporation (NYSE: HAE), a global medical technology company focused on delivering innovative solutions designed to improve patient outcomes, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance for the NexSys PCS® Plasma Collection System with Persona® PLUS technology. Persona PLUS represents the next generation of Haemonetics’ proprietary and patented Persona technology that tailors plasma collections to each donor for improved average plasma volume per donation.
MiRus® Receives FDA 510(k) Clearance for IO™ Expandable Wedge Osteotomy System
ATLANTA, Feb. 23, 2026 /PRNewswire/ — MiRus® today announced FDA 510(k) clearance and commercial launch of the IO™ Expandable Wedge Osteotomy System, an innovative solution designed to bring intraoperative precision and adjustability to foot and ankle osteotomy procedures.
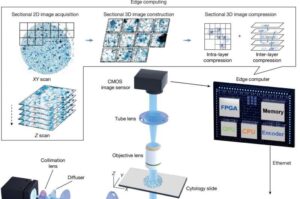
Faster cancer screening? New AI system offers a better way to detect abnormal cells
One way cancer specialists detect the disease is by examining cells and bodily fluids under a microscope, a time-consuming and labor-intensive process called cytology. It involves visually inspecting tens of thousands to one million cells per slide for subtle 3D morphological changes that might signal the onset of cancer. But AI offers an approach that is potentially faster and more accurate.
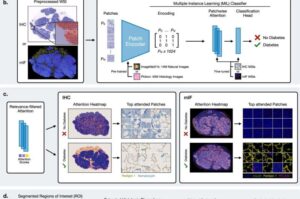
AI decodes tissue characteristics of type 2 diabetes
Researchers from several partner institutions of the German Center for Diabetes Research (DZD) have collaborated with international colleagues to develop a new approach for visualizing subtle tissue changes in the pancreas in type 2 diabetes. The results provide new insights into the development of type 2 diabetes. The study has now been published in Nature Communications.

AI-boosted electronic nose can detect ovarian cancer at an early stage
Using machine learning, an electronic nose can “smell” early signs of ovarian cancer in the blood. The method is precise and, according to the LiU researchers behind the study, it could eventually be used to find many different cancers. The study is published in Advanced Intelligent Systems.

Dry powder inhalers can improve patient outcomes and lower environmental impact
New research from UCLA Health suggests that certain inhalers used to treat chronic obstructive pulmonary disease (COPD) are not only less harmful to the environment but can also lead to slightly better patient outcomes. Inhalers are essential therapies for COPD and other lung conditions, but many commonly used devices rely on propellants that are potent greenhouse gases.
