MedTech News
.................... by Andrew Celentano
New marker identifies patients with advanced prostate cancer more likely to benefit from combination immunotherapy
Researchers with the James P. Allison Institute at The University of Texas MD Anderson Cancer Center have discovered a new gene expression signature within tumors that can help identify patients with metastatic castration-resistant prostate cancer (mCRPC) who are more likely to experience lasting benefits from combined immunotherapy treatment.

Scientists uncover protein that could help failing hearts
Researchers have identified a key protein that may help failing hearts regain function, offering new insight into why some hearts recover while others do not.

AI uses everyday language to make genetic diagnosis easier
The study, conducted by researchers at Baylor College of Medicine and Texas Children’s Hospital, appeared in the American Journal of Human Genetics.
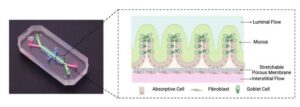
Decoding inflammatory bowel disease—on a chip
In the study, a multi-disciplinary research team led by Wyss Institute at Harvard University Founding Director Donald Ingber, M.D., Ph.D., worked with clinicians at McGill University in Canada and Massachusetts General Hospital who provided IBD and healthy tissue biopsies from the colon region of the same patients to create donor-specific microfluidic organ-on-a-chip (Organ Chip) models of colon that replicate major hallmarks of IBD in vitro in an unprecedented way.

Simple blood test could catch Alzheimer’s and Parkinson’s early by spotting misfolded proteins
A research team led by Professor Klaus Gerwert from Ruhr University Bochum, Germany, has developed just such a blood test. It is based on the immuno-infrared sensor, a novel platform technology to which the Journal of Physical Chemistry B dedicates its cover story in its issue from April 24, 2026.

Healium Announces FDA Registration of Healium Clinical as a Class II 510(k)-Exempt Biofeedback Medical Device
FDA Registration Accelerates Healium Clinical’s Expansion into Healthcare Settings Nationwide

OncoSil Medical secures TGA approval for pancreatic cancer device
The approval is expected to drive increased clinical adoption and support applications for regulatory approval in other markets.

Early warning from low-risk cysts could help catch pancreatic cancer sooner
In a new study, Mass General Brigham investigators showed that patients with low-risk pancreatic cystic lesions (PCLs) have approximately 14 times higher risk of developing pancreatic cancer than the general population.
