MedTech News
.................... by Andrew Celentano
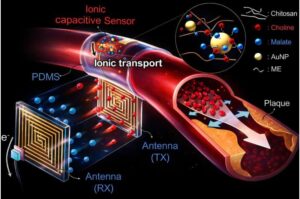
Low-frequency wireless sensor tracks artery stiffening in real time with less interference
A Korean research team has developed a low-frequency-based wireless sensor technology.

ProSomnus gets FDA nod for latest sleep apnea therapy device
ProSomnus announced today that it received FDA 510(k) clearance for its RPMO₂ obstructive sleep apnea (OSA) therapy device.

Beckman Coulter obtains CE mark for bacterial-viral differentiator test
Used with Beckman’s immunoassay analysers, MeMed’s BV test has been demonstrated to reduce unnecessary antibiotic use.
Close, Social Contact Can Shape a Gut Microbiome, Sometimes Making it More Alike
Learn how an isolated but collaborative bird species shows that close social contact can shape our gut microbes in surprising ways.
AI scans 400,000 Reddit posts to flag overlooked GLP-1 side effects
By using AI to analyze more than 400,000 Reddit posts, Penn researchers have identified patient-reported symptoms associated with GLP-1s, the popular weight-loss and diabetes drugs semaglutide and tirzepatide, that may not be fully captured in clinical trials or regulatory documents.
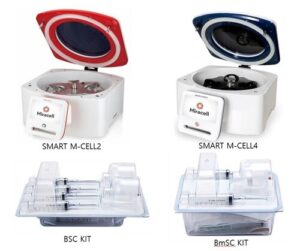
Miracell Receives FDA 510(k) Clearance for SMART M-CELL PRP Concentration System and SMART M-CELL Bone Marrow Concentration System
Blood- and bone marrow-based efficient growth factors extraction system demonstrates substantial equivalence and high cell viability

A new type of electrically driven artificial muscle fiber
Electrofluidic fibers mimic how natural muscle fibers bundle, and could enable compact, silent robotic and prosthetic systems.

InventHelp Inventor Develops Improved Patient Transfer Device (SBT-2234)
PITTSBURGH, April 9, 2026 /PRNewswire/ — “As a patient care coordinator, I thought there could be a better way to lift, transfer, and move a patient,” said an inventor, from Winchester, Calif., “so I invented the IN HOME MANUAL HOIST. My design offers added assistance, and it could be used in place of patient lifts or hydraulic lifts that are bulky and expensive.”
