MedTech News
.................... by Andrew Celentano

Slice and Dice – New Biological Defense Discovered
SNIPE, a newly characterized biological defense system, directly protects bacteria by chopping up invading viral DNA.

Low-Oxygen Levels Boost Wound Healing and Reveal Hidden Regeneration Potential in Mammals
Learn how testing different environmental conditions may unlock mammals’ potential to regrow limbs and what it means for wound healing now.

Modular Medical wins FDA clearance for Pivot tubeless insulin patch pump
Modular Medical (Nasdaq:MODD) announced today that it received FDA 510(k) clearance for its next-generation insulin patch pump.
CorTec gains FDA breakthrough designation for stroke rehabilitation BCI
CorTec’s BCI aims to differentiate itself by offering brain-based communication alongside therapeutic neurostimulation.
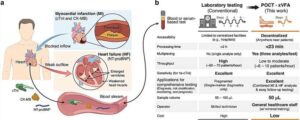
AI-powered portable sensor enables rapid and multiplexed cardiac biomarker testing
Cardiovascular diseases remain the leading cause of death worldwide, accounting for nearly 20 million deaths each year. Rapid diagnosis and risk assessment of cardiac injury are therefore essential for improving patient outcomes.
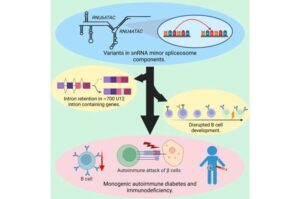
Overlooked non-coding genes cause diabetes in babies, study reveals
Scientists have found new genetic causes for diabetes in babies—in a part of the genome that has historically been overlooked in genetic studies.
Long non-coding RNA may be a promising therapeutic target for cancer
Northwestern Medicine scientists have discovered that a specific long non-coding RNA activates oncogenic signaling pathways in prostate cancer cells and drives tumor progression, underscoring its potential as a therapeutic target, according to a recent study published in Nature Communications.
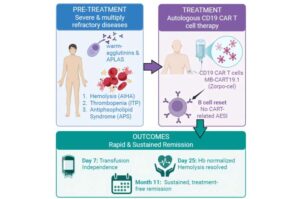
CAR-T therapy drives remission in patient with three autoimmune diseases
For the first time, scientists have used a modern cell therapy called CAR-T to treat a patient with three different life-threatening autoimmune diseases that had resisted years of treatment.
