MedTech News
.................... by Andrew Celentano

LiviWell Secures FDA Clearance for Livi, Introducing a New Category in Post-Intercourse Vaginal Care
MOUNTAIN LAKES, N.J., March 24, 2026 /PRNewswire/ — LiviWell, a women’s health company dedicated to developing innovative solutions for intimate health, today announced that the U.S. Food and Drug Administration (FDA) has cleared Livi, a first-of-its-kind device designed to support vaginal health by absorbing semen and other post-intercourse fluids.
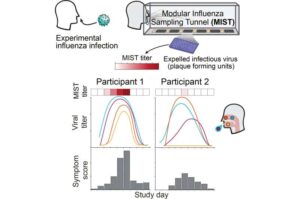
Air-sampling tunnel reveals major person-to-person differences in release of infectious virus
The findings help explain why some people appear far more likely than others to transmit respiratory viruses and offer new insight into how influenza spreads through the air.
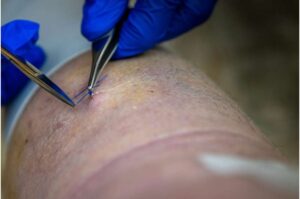
Surgical stitches could release anti-inflammatory drugs at wound sites for weeks
Researchers at Ouachita Baptist University are creating stitches loaded with anti-inflammatory drugs to deliver the medication directly to the injury.

GE HealthCare wins FDA nod for photon-counting CT system
GE HealthCare (Nasdaq:GEHC) announced today that it received FDA 510(k) clearance for its Photonova Spectra solution.
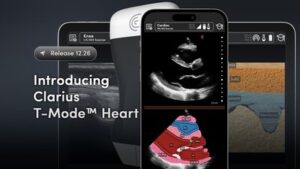
Clarius Unveils T-Mode™ Heart: New AI-Powered Training Tool Empowers Primary Care Physicians to Perform Cardiac Ultrasound Exams
By simplifying complex cardiac anatomy through real-time AI labeling, T-Mode™ Heart removes the training barrier to ultrasound adoption for family physicians and medical students.
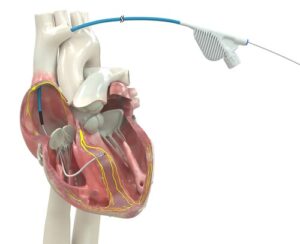
Medtronic OmniaSecure™ defibrillation lead is the first lead of its kind to receive FDA approval for conduction system pacing
Built for high reliability, world’s only lumenless defibrillation lead approved for placement in the left bundle branch area

Hair-thin fiber-optic sensors could detect cancer by reading multiple biomarkers
This breakthrough could lead to next-generation medical tools that track disease, guide treatment and monitor the body in real time

World’s only deuterium-labeled guinea pig helps scientists study metabolism
The method relies on the fact that deuterium, once in the body, participates in biochemical reactions alongside ordinary hydrogen and becomes incorporated into the carbon-hydrogen bonds of organic compounds.
