MedTech News
.................... by Andrew Celentano

DeepHealth Achieves CE Mark for TechLive and Enables AWS Marketplace Deployment
TechLive is a multimodality, vendor-agnostic remote scanning and radiology management solution extending expert oversight across multiple modalities

Abbott earns FDA green light for next-gen CardioMEMS pulmonary artery pressure reader
Abbott (NYSE: ABT)+ today the FDA approved its CardioMEMS Hero next-generation pulmonary artery (PA) pressure reader.

Optellum Secures TGA Approval to Expand AI-Enabled Lung Nodule Solution To Australia
OXFORD, England, Feb. 26, 2026 /PRNewswire/ — Optellum, the leading AI-enabled solution for earlier lung cancer detection, is thrilled to announce that it has received Therapeutic Goods Administration (TGA) class IIb approval in Australia for the Virtual Nodule Clinic (VNC) platform to support early lung cancer diagnosis.
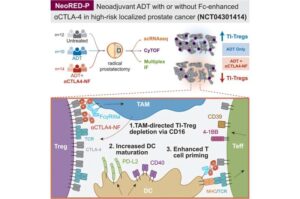
A potential immunotherapy strategy for early-stage prostate cancer
Immunotherapy has been generally ineffective for prostate cancer because the tumors are considered immunologically “cold,” meaning they do not attract enough immune cells to mount a strong attack.

First-ever in-utero stem cell therapy for fetal spina bifida repair shows safe results
A Phase I clinical trial published in The Lancet has shown that combining stem cell therapy with standard fetal surgery before birth is a safe and promising approach to treat myelomeningocele, a severe form of spina bifida.
Laser Therapy Boosts Survival in Treating Brain Cancer, With Nearly Half Alive at 18 Months
Learn how a new laser-based therapy is giving patients with aggressive brain cancer a stronger chance at survival.
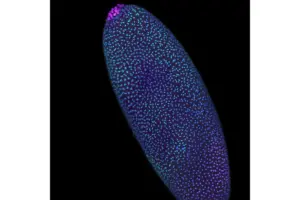
Imaging Technique Reveals DNA’s Hidden Shape in the Earliest of Embryos
Learn more about Pico-C, a tool that helps reveal the genome’s structure during the first days of life.
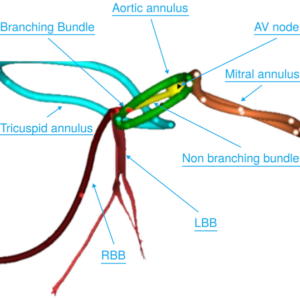
Cara Medical Receives FDA 510(k) Clearance for the CARA System for Noninvasive CTA based Cardiac Conduction System Visualization
TORTOLA, British Virgin Islands, Feb. 25, 2026 /PRNewswire/ — Cara Medical Ltd. today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for the CARA System, a computed tomography angiography (CTA)-based platform that provides noninvasive, patient-specific three-dimensional (3D) visualization of the cardiac conduction system.
