MedTech News
.................... by Andrew Celentano
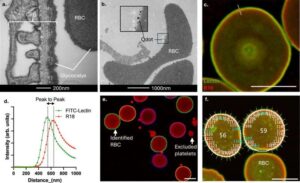
Blood test enables earlier detection of heart and kidney disease
A new way to detect the onset of heart and kidney disease far earlier than previously possible has been discovered by scientists. The breakthrough, published today in Nature Communications, reveals a novel method for identifying damage to the lining of microscopic blood vessels.

AI-powered handheld microscope aims to spot cancer earlier
Researchers at Rice University and The University of Texas MD Anderson Cancer Center have developed a compact, artificial intelligence-powered imaging device that could transform how clinicians detect cancer. The technology, which aims to bring high-resolution, real-time diagnostics directly to the point of care, was recently described in a paper published in the Proceedings of the National Academy of Sciences.
Successfully treated acute myeloid leukemia patients may hold the key to new CAR T cell therapy
Now, a team of researchers from Memorial Sloan Kettering Cancer Center (MSK) has developed a new type of CAR T cell therapy that targets a protein found almost exclusively on leukemia cells and not on healthy cells.
Gene-edited stem cell transplant shows promise for aggressive blood cancers
Researchers at Washington University School of Medicine in St. Louis, showed that a stem cell transplant in which the donor cells have been genetically engineered to remove a particular protein helps prevent toxic side effects and potentially improves the effectiveness of therapies given after a transplant to help prevent cancer recurrence.
Bayesian Health secures first FDA clearance for AI-driven continuous sepsis monitor
The US Food and Drug Administration (FDA) has cleared Bayesian Health’s continuous artificial intelligence (AI)-driven sepsis monitor, making the device the first of its kind to achieve this milestone.
Siemens Healthineers wins FDA clearance for 6 new interventional imaging systems
Siemens Healthineers announced today that the FDA cleared six new systems within its Artis interventional imaging portfolio.
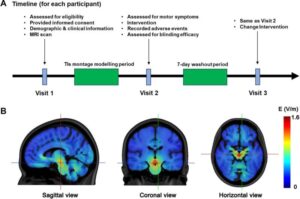
Noninvasive deep brain stimulation technique shows early promise for treating Parkinson’s disease
A novel, noninvasive brain stimulation approach—known as transcranial temporal interference stimulation (TIs)—may offer a new way to treat motor symptoms in Parkinson’s disease without the need for surgery, according to a pilot study appearing in eBioMedicine. The technique, which uses overlapping electrical currents to selectively target deep brain regions, significantly improved movement in patients compared with a sham treatment when targeting the subthalamic nucleus.

Cardiac MRI and blood markers sharpen hypertrophic cardiomyopathy risk prediction
Findings from a new study have identified a new model for predicting outcomes for hypertrophic cardiomyopathy (HCM), a heart condition with a prevalence of one in 500 people and a frequent cause of sudden cardiac death.
