Breast tumors use sugar coating to evade immunity, opening potential immunotherapy path
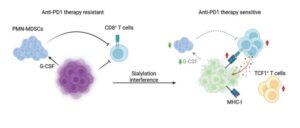
An international research team led by the Medical University of Vienna has now identified a previously underestimated mechanism by which breast tumors evade the immune system.